Why Your Doctor Recommends Circulating Tumor DNA (ctDNA)? Everything You Should Know
Abstract
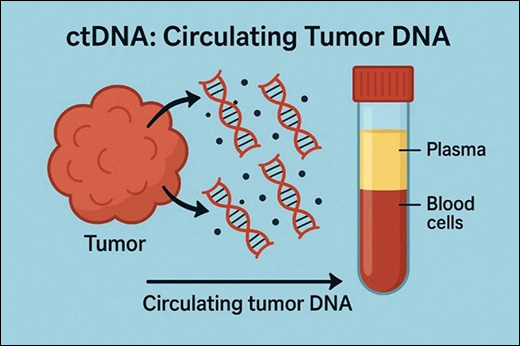
Tumor cells continuously shed nucleic acid fragments into the bloodstream. These fragments form part of circulating cell-free DNA (cfDNA), a heterogeneous mixture of DNA derived from both normal and malignant cells. The tumor-derived portion of this pool is known as circulating tumor DNA. Unlike traditional tissue biopsy, which samples a single tumor site at a single time point, ctDNA reflects molecular information from multiple tumor regions simultaneously. Because ctDNA can be obtained through a simple blood sample, it represents a non-invasive method for evaluating tumor biology and monitoring disease progression over time. This approach is often described as a “liquid biopsy,” highlighting its role as an alternative to invasive tissue sampling. This article presents a comprehensive, research-based overview of ctDNA biology, mechanisms of release, laboratory detection methods, clinical applications, interpretation principles, advantages, limitations, and emerging roles in precision medicine.
Introduction
Circulating tumor DNA (ctDNA) is a fraction of cell-free DNA released into the bloodstream from tumor cells undergoing apoptosis, necrosis, or active secretion. As a minimally invasive biomarker, ctDNA provides real-time molecular insight into tumor genetics, disease burden, treatment response, and emergence of resistance mechanisms. Technological advances in high-sensitivity molecular detection have enabled ctDNA analysis to become a powerful tool in oncology research and clinical decision-making.
Biological Basis of ctDNA
Origin And Release Mechanisms
Tumor cells release DNA fragments into circulation through several biological processes:
- Apoptosis resulting in fragmentation of nuclear DNA
- Necrosis associated with rapid tumor growth and hypoxia
- Active secretion via extracellular vesicles
- Cellular turnover within tumor microenvironment
These DNA fragments enter the bloodstream and become detectable within plasma. ctDNA is typically highly fragmented, reflecting nucleosomal DNA packaging patterns.
Molecular Characteristics
ctDNA carries tumor-specific genetic alterations, which may include:
- Point mutations
- Insertions and deletions
- Copy number variations
- Structural rearrangements
- Methylation changes
Because these alterations arise from malignant transformation, ctDNA provides molecular information directly linked to tumor genetics.
Relationship Between ctDNA and Tumor Biology
The quantity of ctDNA in circulation depends on multiple biological factors:
- Tumor size and growth rate
- Degree of vascularization
- Cellular turnover
- Location of metastases
- Treatment-induced cell death
Higher tumor burden generally correlates with increased ctDNA concentration, although release patterns vary among tumor types and individuals.
Laboratory Measurement And Detection Methods
Sample Collection
ctDNA analysis is performed using plasma obtained from venous blood. Specialized collection tubes are often used to stabilize nucleic acids and prevent contamination from leukocyte DNA.
Processing steps include:
- Blood collection
- Plasma separation through centrifugation
- DNA extraction
- Molecular analysis
Strict pre-analytical handling is essential to ensure accurate measurement.
Analytical Techniques
Digital Polymerase Chain Reaction (dPCR)
This method partitions DNA into numerous reactions, allowing detection of rare tumor-specific mutations with high sensitivity.
Next-Generation Sequencing (NGS)
NGS enables simultaneous analysis of multiple genetic regions and provides comprehensive tumor genomic profiling.
Targeted Mutation Analysis
Assays designed to detect known tumor mutations provide rapid and focused molecular evaluation.
Methylation-Based Detection
- Epigenetic patterns specific to tumor DNA can be identified using specialized assays.
- Each method varies in sensitivity, breadth of detection, and clinical application.
Quantitative And Qualitative Interpretation
Presence or Absence of ctDNA
Detection of tumor-specific DNA indicates presence of malignant cellular activity. Absence of detectable ctDNA does not exclude disease, particularly in early-stage tumors with low DNA shedding.
Quantitative Levels
Changes in ctDNA concentration over time provide insight into disease dynamics:
- Rising levels suggest increasing tumor activity
- Declining levels indicate treatment response
- Stable levels suggest disease equilibrium
Molecular Profiling
Identification of specific genetic alterations supports understanding of tumor behavior and potential therapeutic strategies.
Clinical Applications
Early Cancer Detection
ctDNA analysis has been investigated as a tool for identifying malignancy before clinical symptoms appear. Detection of tumor-specific mutations or methylation patterns may enable early disease recognition.
Molecular Characterization of Tumors
ctDNA allows assessment of tumor genetic profile without invasive biopsy. This is particularly valuable when tissue sampling is difficult or unsafe.
Monitoring Treatment Response
Therapeutic interventions often cause tumor cell death, leading to temporary increases followed by declines in ctDNA levels. Serial monitoring provides dynamic information regarding treatment effectiveness.
Detection of Minimal Residual Disease
After treatment, persistent ctDNA may indicate microscopic residual disease not detectable through imaging.
Identification of Resistance Mechanisms
Tumor evolution under treatment pressure may produce new genetic alterations. ctDNA analysis can reveal these changes and inform therapeutic adjustments.
Surveillance for Recurrence
Reappearance of ctDNA following treatment may precede clinical or radiologic evidence of disease recurrence.
Comparison With Conventional Diagnostic Approaches
Traditional tissue biopsy provides structural and histological information but has limitations:
- Invasive procedure
- Sampling bias
- Limited ability for repeated monitoring
- Single-site evaluation
ctDNA analysis complements these methods by offering:
- Non-invasive sampling
- Real-time molecular assessment
- Systemic tumor representation
- Feasibility of serial monitoring
However, ctDNA does not replace histopathological diagnosis and must be interpreted within a comprehensive clinical framework.
Factors Affecting ctDNA Detection
Several variables influence detectability and interpretation:
- Tumor stage and burden
- Biological shedding rate
- Clearance of circulating DNA
- Analytical sensitivity of assay
- Pre-analytical sample handling
Understanding these factors is essential for accurate clinical interpretation.
Advantages Of Ctdna Analysis
- Minimally invasive
- Reflects tumor heterogeneity
- Enables longitudinal monitoring
- Provides molecular-level insight
- Detects genetic changes dynamically
- Supports precision medicine approaches
Limitations And Challenges
- Low abundance in early-stage disease
- Technical variability across platforms
- Lack of universal standardization
- Interpretation requires clinical correlation
- Potential false-negative results in low-shedding tumors
- Biological complexity of cfDNA mixture
Ongoing research aims to address these challenges through improved detection technologies and standardized methodologies.
Role In Precision Medicine
ctDNA analysis aligns with personalized medicine by enabling treatment decisions based on tumor-specific molecular features. By capturing dynamic genetic changes, ctDNA provides insight into tumor evolution and therapeutic vulnerability. This supports individualized management strategies tailored to molecular disease characteristics.
Research Developments
Recent advances in molecular diagnostics have expanded the potential applications of ctDNA. Areas of active investigation include:
- Multi-cancer early detection platforms
- Integration with imaging and clinical data
- Epigenetic biomarker profiling
- Monitoring immune therapy response
- Predictive modeling of disease progression
These developments continue to refine the clinical utility of ctDNA as a biomarker of systemic tumor activity.
Integrative Perspective On Cellular Stability And Hepatic Health
Maintenance of cellular homeostasis and metabolic balance is fundamental to preserving tissue integrity. Traditional hepatoprotective botanical approaches emphasize support of antioxidant defense systems, metabolic regulation, and cellular resilience. From a biomedical perspective, several herbs demonstrate properties that support hepatic functional stability under metabolic stress.
Bhumi Amalaki (Phyllanthus niruri)
Bhumi Amalaki (Phyllanthus niruri) contains lignans and polyphenols that support antioxidant defenses and hepatocyte membrane integrity. Experimental findings suggest modulation of oxidative stress pathways involved in cellular injury.
Kalmegha (Andrographis paniculata)
Kalmegha (Andrographis paniculata) is rich in diterpenoid compounds with anti-inflammatory and hepatocyte-protective properties. Supports metabolic balance and cellular adaptation to stress.
Kutki (Picrorhiza kurroa)
Kutki (Picrorhiza kurroa) contains iridoid glycosides associated with hepatocyte stabilization and reduction of oxidative injury, supporting maintenance of cellular homeostasis.
Bhringaraja (Eclipta alba)
Bhringaraja (Eclipta alba) provides antioxidant compounds that support hepatocyte regeneration and preservation of structural integrity.
Punarnava (Boerhavia diffusa)
Punarnava (Boerhavia diffusa) supports metabolic regulation and microcirculatory balance while contributing to maintenance of hepatic tissue function.
These botanicals are considered supportive for maintaining physiological liver function but do not substitute clinical evaluation or oncologic care.
Conclusion
Circulating tumor DNA represents a transformative advancement in molecular diagnostics, offering a non-invasive window into tumor genetics and disease dynamics. By capturing tumor-derived genetic material in the bloodstream, ctDNA enables real-time monitoring of malignant activity, treatment response, and molecular evolution. Although technical and biological challenges remain, ongoing research continues to expand its clinical relevance. When integrated with conventional diagnostic methods and clinical assessment, ctDNA analysis contributes to a comprehensive and dynamic understanding of cancer biology and supports the advancement of precision medicine.



